
NMCONNECT team
Co-team leaders: Bertrand Fontaine & Laure Strochlic
Our laboratory aims to understand the cellular and molecular mechanisms underlying neuromuscular synapse assembly and maintenance in physiological and pathological conditions.
The Neuromuscular junction (NMJ) is the contact zone between motor neurons and skeletal muscles. This synapse drives the precise initiation and control of motion. Therefore, much of our behavior and wellness relies on the appropriate functioning of this specialised structure. Neuromuscular transmission deficiency occurs in a large array of rare human diseases including channelopathies, congenital or acquired myasthenia and amyotrophic lateral sclerosis. Most of these pathologies are untreatable and life threatening with devastating economic and societal consequences in terms of loss of quality of life and of the burden of disability. The patients display complex clinical phenotypes mainly characterized by a profound muscle weakness and loss of mobility.
We combine unbiased screens and a large array of functional assays including quantitative morphological imaging, behavioral analysis and electrophysiology using mouse models and/or human-derived specimens to explore the complexity of the trans-synaptic mechanisms controlling neuromuscular connectivity. Our overarching goal is to improve physiopathological knowledge that can be used not only for molecular diagnosis and genetic counselling of families affected with the diseases of interest, but also to design new targets of therapeutic interest.
To achieve this goal, our team is built on an organization that favors tight interactions between practicing neurologists from the Paris Est French reference center for neuromuscular diseases and researchers/research assistants together with a large network of national/international collaborations to share knowledge and expertise.
We defined three main aims of research
- Characterize the molecular determinants underlying NMJ assembly and maintenance (PI: Laure Strochlic)
We have recently identified a new trans-synaptic pathway at the NMJ and developed innovative tools using mouse genetics to dissect its molecular characteristics.

- Understand how disruption of nerve/muscle communication leads to neuromuscular diseases such as myasthenia, amyotrophic lateral sclerosis and aged-related muscle wasting (PI: Gaelle Bruneteau).
Thanks to our clinical expertise and our national networks, we analyze the physiopathological mechanisms underlying the studied diseases in patients.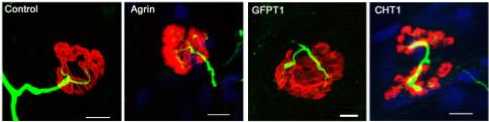
- Modulate trans-synaptic function to restore appropriate synaptic connectivity in a pathological context as a basis for therapeutic interventions (PI: Bertrand Fontaine/Laure Strochlic).
We develop innovative pharmacological or genetic strategies that promote trans-synaptic communication and nerve/muscle attachment to mitigate NMJ disease symptoms to ultimately prevent or compensate the progression of the loss of motor function in neuromuscular diseases.
Key words: Neuromuscular Junction, Neuromuscular disorders, disease modeling mouse models, human derived specimens, quantitative imaging, therapeutic strategies.
Major collaborations
Rozen Le Panse and Sonia Berrih-Aknin, Myology research center (France).
Arnaud Ferry, Myology research center (France).
Nathalie Sans and Mireille Montcouquiol, Magendie Neurocenter (France).
Laurent Schaeffer, NeuroMyoGene Institute, (France).
Hanns Lochmüller, The Children’s Hospital of Eastern Ontario (Canada).
Markus Ruëgg, Biozentrum (Switzerland).
Anna Punga, Uppsala University, (Sweden).

Team composition
Group Leaders: Bertrand Fontaine (PU-PH, SU/APHP) and Laure Strochlic (Inserm, CR1)
Research team
Céline Buon (IE, Inserm CDD)
Marius Halliez (PhD student, SU)
Céline Douarre (Post-doc, Inserm, CDD)
Lucile Grimbert (Post-doc, Inserm, CDD)
Antony Grondin (Engineer, Inserm, CDD))
Part-time clinicians
Gaëlle Bruneteau (PH, APHP, 20%)
Damien Sternberg (PH, APHP, 20%)
Arnaud Isapof (PH, APHP, 20%)
Savine Vicart (PH, APHP, 20%)
Karine Viala (PH, APHP, 20%)
Last publications
- Boëx M, Cottin S, Halliez M, Bauché S, Buon C, Sans N, Montcouquiol M, Molgó J, Amar M, Ferry A, Lemaitre M, Rouche A, Langui D, Baskaran A, Fontaine B, Messéant J, Strochlic L. The cell polarity protein Vangl2 in the muscle shapes the neuromuscular synapse by binding to and regulating the tyrosine kinase MuSK. Sci Signal. 2022 May 17;15(734):eabg4982. doi: 10.1126/scisignal.abg4982. Epub 2022 May 17. PMID: 35580169.
- L. Strochlic. Entre muscle et nerf : comprendre la jonction neuromusculaire pour mieux comprendre le mouvement. The Conversation, July 2022.
- C. Offord, L. Strochlic. A Novel Player at the Neuromuscular Junction. The Scientist, Sept 2022.
- Savvatis K, Vissing CR, Klouvi L, Florian A, Rahman M, Béhin A, Fayssoil A, Masingue M, Stojkovic T, Bécane HM, Berber N, Mochel F, Duboc D, Fontaine B, Krett B, Stalens C, Lejeune J, Pitceathly RDS, Lopes L, Saadi M, Gossios T, Procaccio V, Spinazzi M, Tard C, De Groote P, Dhaenens CM, Douillard C, Echaniz-Laguna A, Quinlivan R, Hanna MG, Yilmaz A, Vissing J, Laforêt P, Elliott P, Wahbi K. Cardiac Outcomes in Adults With Mitochondrial Diseases. J Am Coll Cardiol. 2022 Oct 11;80(15):1421-1430. doi: 10.1016/j.jacc.2022.08.716. PMID: 36202532.
- Villar-Quiles RN, Sternberg D, Tredez G, Beatriz Romero N, Evangelista T, Lafôret P, Cintas P, Sole G, Sacconi S, Bendahhou S, Franques J, Cances C, Noury JB, Delmont E, Blondy P, Perrin L, Hezode M, Fournier E, Fontaine B, Stojkovic T, Vicart S. Phenotypical variability and atypical presentations in a French cohort of Andersen-Tawil syndrome. Eur J Neurol. 2022 Aug;29(8):2398-2411. doi: 10.1111/ene.15369. Epub 2022 May 4. PMID: 35460302.
- Lévêque N, Slis A, Lancia L, Bruneteau G, Fougeron C. Acoustic Change Over Time in Spastic and/or Flaccid Dysarthria in Motor Neuron Diseases. J Speech Lang Hear Res. 2022 May 11;65(5):1767-1783. doi: 10.1044/2022_JSLHR-21-00434. Epub 2022 Apr 12. PMID: 35412848
- Le Gall L, Duddy WJ, Martinat C, Mariot V, Connolly O, Milla V, Anakor E, Ouandaogo ZG, Millecamps S, Lainé J, Vijayakumar UG, Knoblach S, Raoul C, Lucas O, Loeffler JP, Bede P, Behin A, Blasco H, Bruneteau G, Del Mar Amador M, Devos D, Henriques A, Hesters A, Lacomblez L, Laforet P, Langlet T, Leblanc P, Le Forestier N, Maisonobe T, Meininger V, Robelin L, Salachas F, Stojkovic T, Querin G, Dumonceaux J, Butler Browne G, González De Aguilar JL, Duguez S, Pradat PF. Muscle cells of sporadic amyotrophic lateral sclerosis patients secrete neurotoxic vesicles. J Cachexia Sarcopenia Muscle. 2022 Apr;13(2):1385-1402. doi: 10.1002/jcsm.12945. Epub 2022 Feb 22. PMID: 35194965; PMCID: PMC8978001
- Querin G, Lenglet T, Debs R, Stojkovic T, Behin A, Salachas F, Le Forestier N, Amador MDM, Bruneteau G, Laforêt P, Blancho S, Marchand-Pauvert V, Bede P, Hogrel JY, Pradat PF. Development of new outcome measures for adult SMA type III and IV: a multimodal longitudinal study. J Neurol. 2021 Jan 2. doi: 10.1007/s00415-020-10332-5
- Herbst R, Koneczny I, Lochmüller H and Strochlic L. Editorial: Molecular Mechanisms Underlying Assembly and Maintenance of the Neuromuscular Junction. (2021) Front. Mol. Neurosci. 14:797832. doi: 10.3389/fnmol.2021.797832
- Porcher R, Desguerre I, Amthor H, Chabrol B, Audic F, Rivier F, Isapof A, Tiffreau V, Campana-Salort E, Leturcq F, Tuffery-Giraud S, Ben Yaou R, Annane D, Amédro P, Barnerias C, Bécane HM, Béhin A, Bonnet D, Bassez G, Cossée M, de La Villéon G, Delcourte C, Fayssoil A, Fontaine B, Godart F, Guillaumont S, Jaillette E, Laforêt P, Leonard-Louis S, Lofaso F, Mayer M, Morales RJ, Meune C, Orlikowski D, Ovaert C, Prigent H, Saadi M, Sochala M, Tard C, Vaksmann G, Walther-Louvier U, Eymard B, Stojkovic T, Ravaud P, Duboc D, Wahbi K. Association between prophylactic angiotensin-converting enzyme inhibitors and overall survival in Duchenne muscular dystrophy-analysis of registry data. Eur Heart J. 2021 May 21;42(20):1976-1984. doi: 10.1093/eurheartj/ehab054. PMID: 33748842.
- Delorme C, Houot M, Rosso C, Carvalho S, Nedelec T, Maatoug R, Pitron V, Gassama S, Sambin S, Bombois S, Herlin B, Ouvrard G, Bruneteau G, Hesters A, Gales AZ, Millet B, Lamari F, Lehericy S, Navarro V, Rohaut B, Demeret S, Maisonobe T, Yger M, Degos B, Mariani LL, Bouche C, Dzierzynski N, Oquendo B, Ketz F, Nguyen AH, Kas A, Lubetzki C, Delattre JY, Corvol JC; CoCo-Neurosciences Study Group.The wide spectrum of COVID-19 neuropsychiatric complications within a multidisciplinary centre. Brain Commun. 2021 Jun 17;3(3):fcab135. doi: 10.1093/braincomms/fcab135. eCollection 2021.PMID: 34401746
- Carvalho K, Martin E, Ces A, Sarrazin N, Lagouge-Roussey P, Nous C, Boucherit L, Youssef I, Prigent A, Faivre E, Eddarkaoui S, Gauvrit T, Vieau D, Boluda S, Huin V, Fontaine B, Buée L, Delatour B, Dutar P, Sennlaub F, Guillonneau X, Blum D, Delarasse C; NeuroCEB Brain Bank. P2X7-deficiency improves plasticity and cognitive abilities in a mouse model of Tauopathy. Prog. Neurobiol. 2021 Nov;206:102139. doi: 10.1016/j.pneurobio.2021.102139. Epub 2021 Aug 12.PMID: 34391810
- Stalens C, Motté L, Béhin A, Ben Yaou R, Leturcq F, Bassez G, Laforêt P, Fontaine B, Ederhy S, Masingue M, Saadi M, Louis SL, Berber N, Stojkovic T, Duboc D, Wahbi K. Improved Cardiac Outcomes by Early Treatment with Angiotensin-Converting Enzyme Inhibitors in Becker Muscular Dystrophy. J Neuromuscul Dis. 2021;8(4):495-502. doi: 10.3233/JND-200620.PMID: 33814458
- Desaphy JF, Altamura C, Vicart S, Fontaine B. Targeted Therapies for Skeletal Muscle Ion Channelopathies: Systematic Review and Steps Towards Precision Medicine. J Neuromuscul Dis. 2021;8(3):357-381. doi: 10.3233/JND-200582.PMID: 33325393.
- Vicart S, Franques J, Bouhour F, Magot A, Péréon Y, Sacconi S, Nadaj-Pakleza A, Behin A, Zahr N, Hézode M, Fournier E, Payan C, Lacomblez L, Fontaine B. Efficacy and safety of mexiletine in non-dystrophic myotonias: A randomised, double-blind, placebo-controlled, cross-over study. Neuromuscul Disord. 2021 Nov;31(11):1124-1135. doi: 10.1016/j.nmd.2021.06.010.
- Gómez-Fernández P, Lopez de Lapuente Portilla A, Astobiza I, Mena J, Urtasun A, Altmann V, Matesanz F, Otaegui D, Urcelay E, Antigüedad A, Malhotra S, Montalban X, Castillo-Triviño T, Espino-Paisán L, Aktas O, Buttmann M, Chan A, Fontaine B, Gourraud PA, Hecker M, Hoffjan S, Kubisch C, Kümpfel T, Luessi F, Zettl UK, Zipp F, Alloza I, Comabella M, Lill CM, Vandenbroeck K. The Rare IL22RA2 Signal Peptide Coding Variant rs28385692 Decreases Secretion of IL-22BP Isoform-1, -2 and -3 and Is Associated with Risk for Multiple Sclerosis. Cells. 2020 Jan 10;9(1):175. doi: 10.3390/cells9010175. PMID: 31936765; PMCID: PMC7017210.

