 Team leader : Jean-Yves Hogrel
Team leader : Jean-Yves Hogrel
Objectives
The Neuromuscular Physiology and Evaluation Laboratory (NeuPEL) is part of the Institute of Myology’s Neuromuscular Investigation Center. The main goal of this laboratory is to understand neuromuscular system changes and adaptations and to assess the effects of therapeutic interventions. It is a multidisciplinary laboratory with multiple areas of expertise in the fields of physiology, health, technology, rehabilitation science and data science. In this context, innovative approaches are being developed for the non-invasive assessment of the neuromuscular system, motor performance and physical activity. These approaches are being particularly investigated in patients with neuromuscular diseases in the context of clinical research protocols to understand disease progression and to evaluate the efficacy of innovative therapies. These approaches are also being investigated in other clinical situations with a neuromuscular impact, such as ageing, chronic diseases, intensive care and exercise. The laboratory is also conducting prospective studies regarding the effects of physical activity and the use of innovative assistance devices in patients with neuromuscular disease. The NeuPEL laboratory is involved in the clinical diagnosis of patients presenting rare metabolic diseases. Finally, the NeuPEL laboratory is also providing services and consultancy to academic and industrial collaborators (design assistance, clinical metrology, training, patient assessment).
The NeuPEL laboratory is attached to the Physiology Pathophysiology Doctorate School. (ED394, Sorbonne université).

Team members
- Jean-Yves Hogrel, PhD, HDR, MSc, Biomedical engineer, Team leader
- Valérie Decostre, PhD, Physiotherapist, Researcher
- Simone Birnbaum, PhD, Physiotherapist, Researcher
- Isabelle Ledoux, MSc, Biomedical engineer
- Romain Feigean, PhD, Researcher
- Thomas Marques, Research Engineer
- Stéphanie Berthier, Ingénieur qualité
- Audrey El Kaim, MSc, Physiotherapist
- Pauline Santmarty, Physiotherapist
- Marjorie Banos, MSc, Physiotherapist
- Ghida Trad, MSc, Physiotherapist, PhD student
Contact
Tel : 01 42 16 58 79
Email : eval@institut-myologie.org
Innovative approaches to the non-invasive assessment of the neuromuscular system
Assessment of assistance devices – AssistMyo
Exercise and physical activity in the neuromuscular diseases – ExMyo
Clinical diagnosis activities
Involvement in clinical research
Expertise et services
Patents
Last publications
Innovative approaches to the non-invasive assessment of the neuromuscular system
Shortly after the creation of the laboratory, it quickly became clear that commercial solutions to neuromuscular patient assessment had prohibitive limitations or simply did not exist at all. In 1999, we participated in the regional assessment of one of the first gene therapies for Duchenne muscular dystrophy (Transgene trial). A plasmid was injected into the radial wrist extensor muscle. There were no devices in existence to measure the wrist extension torque. We therefore developed a specific dynamometer (MyoWrist) to optimally assess this innovative treatment. There has been an explosion in the number of therapeutic approaches to neuromuscular disease in the last 10 years. The choice of primary and secondary outcome measures used is crucial in assessing potential treatment benefits. Therefore, one of our objectives is to generate the knowledge that will help to choose the best outcome measures to assess the neuromuscular system and motor performance in a given clinical situation. The development of non-invasive measurement is also one of the key laboratory research programme objectives. Our current approaches rely mainly on a combination of specific devices, inertial measurements, electrophysiological measurements and multi-parametric ultrasound imaging (shear wave elastography, ultrafast imaging). Validation of the different approaches and devices that we are developing, and the development of normative data, are together a key activity and area of expertise of the laboratory.
Devices to assess neuromuscular function – MyoTools
MyoTools are a collection of devices developed in the laboratory, primarily to assess muscle strength. These devices are particularly suited to assessing adults and children with neuromuscular disease, but also have a number of applications in other clinical fields (ageing, cardiorespiratory and metabolic diseases, etc.). In addition to the devices themselves, MyoTools encompasses a set of standardised procedures, a quality approach guaranteeing the best possible clinical metrology, and the provision of training. MyoTools are currently being used in many clinical trials worldwide.
A number of the MyoTools are manufactured and distributed by our partners.
MyoTools
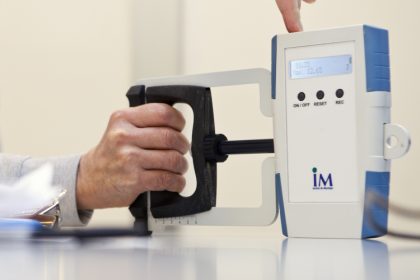
MyoGrip- A high-precision dynamometer to assess grip strength (patented)
MyoGrip is an electronic dynamometer specially designed to measure grip strength under isometric conditions and suited to very weak patients. MyoGrip was developed to meet the need for a quantified and rigourous follow up of grip strength, especially in therapeutic trials. MyoGrip measures forces of between 0 and 90 kg with a resolution of 10 g and an accuracy of 50 g. The handle width is adjustable.
It is manufactured and distributed by Ateliers Laumonier (contact@laumonier.fr).

 MyoPinch – A high-precision dynamometer to assess thumb-index finger pinch strength (patented)
MyoPinch – A high-precision dynamometer to assess thumb-index finger pinch strength (patented)
MyoPinch is an electronic dynamometer specially designed to measure thumb-index finger pinch strength and suited to very weak patients. MyoPinch was developed to meet the need for a quantified and rigourous follow up of thumb-index finger pinch strength, especially in therapeutic trials. MyoPinch measures forces of between 0 and 18 kg with a resolution of 1 g and an accuracy of 10 g.
It is manufactured and distributed by Ateliers Laumonier (contact@laumonier.fr).
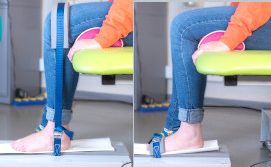 MyoAnkle – A dynamometer to assess ankle flexion and extension strength
MyoAnkle – A dynamometer to assess ankle flexion and extension strength
MyoAnkle is an electronic dynamometer specially designed to measure ankle flexion and extension strength in a reliable and reproducible manner. The range of MyoAnkle measurements is suited to patients with muscular diseases, as well as to healthy individuals.
It is manufactured and distributed by Ateliers Laumonier (contact@laumonier.fr).
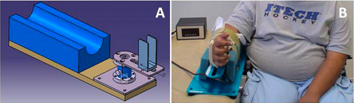 MyoWrist – A dynamometer to assess wrist flexion and extension strength
MyoWrist – A dynamometer to assess wrist flexion and extension strength
MyoWrist is an electronic dynamometer specially designed to measure wrist flexion and extension strength in a reliable and reproducible manner. The range of MyoWrist measurements is suited to patients with muscular diseases (0.1 N m to 5 N m), as well as to healthy individuals (25 N m). The ergonomics of the device are also suited to use by wheelchair patients. The MyoWrist is adjustable, allowing measurements to be performed on children or adults.

 MyoQuad – A dynamometer to assess knee extensor strength (patented)
MyoQuad – A dynamometer to assess knee extensor strength (patented)
MyoQuad is an electronic dynamometer specially designed to measure knee extensor muscle strength and suited to very weak patients. MyoQuad was designed to be used equally in a hospital, home or office-based consultation setting. The idea is that it can be hooked onto the leg of any medical bed or chair, whether round or rectangular. The dynamometer, which is hooked at one end to a fixed support and at the other to the patient’s ankle, is connected by Bluetooth to a computer, which allows measurements to be visualised in real-time, using a dedicated application. It is able to detect 10 g variations in force with an accuracy of 50 g.
It is manufactured and distributed by Ateliers Laumonier (contact@laumonier.fr).

 MoviPlate – A device to assess distal motor function of the upper limb (patented)
MoviPlate – A device to assess distal motor function of the upper limb (patented)
MoviPlate is an instrument to assess motor function of the upper limbs, particularly suited to non-mobile patients. It makes it possible to assess the efficacy of therapies on distal motor function of the upper limbs in the context of therapeutic trials.
It is manufactured and distributed by Valotec (contact@valotec.fr).
Protocols and Publications in which MyoTools have been used
Among the tools developed by the laboratory, the MyoGrip and MyoPinch dynamometers have been distributed and used in several natural history studies (DMD, SMA, GNE, IBM, CNM, MTM, LGMD, GSDIII) and therapeutic trials (Solid, Dynacure, Antisense, Généthon). They are currently the most sensitive and accurate devices of this type on the market. They have given rise to many publications, the most iconic of which relate to DMD (Servais et al, 2013; Seferian et al, 2015; Hogrel et al, 2016, 2020; Ricotti et al, 2016, 2019) and SMA (Seferian et al, 2015; Chabanon et al, 2018; Querin et al, 2021; Annoussamy et al, 2021).
MyoTools training
Have you acquired one of our muscle assessment devices? Training in the use of these devices, by videoconference or on-site, is available on request, as well as a user manual and data collection sheet, at myohelp@institut-myologie.org.
Quality control
Quality control of the data collected by the tools developed at the laboratory can be performed in the context of clinical research protocols. The aim is to be able to guarantee the quality of the collected data. For the MyoGrip, MyoPinch and Moviplate devices, it is possible to record the readings using a dedicated software application. This allows the recorded signals to be re-read subsequently, if there is any doubt about the reliability of the results.
Normative data – the MyoTools application- MyoApp
Normative data are obtained from data collected among children and adults in a good state of health. Predictive models are then established, allowing the normal value expected for a particular individual, according to their age, height, weight, sex or other variables, to be calculated. For example, the application below makes it possible to quickly calculate the predicted value for measured force with MyoTools.
Disclaimer
The information, including but not limited to, text, graphics, images and other material contained on this website are for informational purposes only. It is not intended to be a substitute for professional medical advice, diagnosis or treatment. Always seek the advice of your physician or other qualified health care provider with any questions you may have regarding a medical condition or treatment and before undertaking a new health care regimen, and never disregard professional medical advice or delay in seeking it because of something you have read on this website.
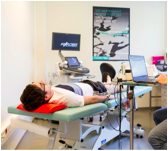 Assessment of regional muscle volume using bio-impedancemetry – ElecMyo
Assessment of regional muscle volume using bio-impedancemetry – ElecMyo
We are developing new approaches, based on the study of the electrical properties of biological tissues, to quantify regional muscle mass (for example, the thigh). This work involves the development of devices (e.g. skin-electrode interface), in silico models, and the collection of data among healthy volunteers in a variety of physiological contexts (immobilisation, training, etc.), and among patients with neuromuscular diseases or diseases that have an impact on the muscles. This work is being conducted in collaboration with the Institute of Myology’s NMR laboratory, the Department of Neuromyology, and the APHP-SU Department of Internal Medicine..
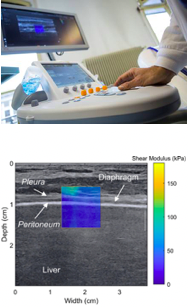 Multi-parametric ultrasound of locomotion and respiratory muscles – RespiMyo
Multi-parametric ultrasound of locomotion and respiratory muscles – RespiMyo
We are developing new approaches based on multi-parametric ultrasound, to characterise muscle structure and function. This work includes, in particular, the use of ultrafast plane wave imaging, deformation imaging, and shear wave elastography. Within this field, the highly ambitious RespiMyo project is focusing on the application of multi-parametric ultrasound to the diaphragm, in order to be able to offer specific and non-invasive alternatives to assess diaphragm dysfunction associated with neuromuscular diseases, respiratory diseases or diseases resulting from mechanical ventilators in intensive care. This work is being conducted in collaboration with the Institute of Myology’s NMR laboratory, the BIOMAPS laboratory (Paris-Sud University – CEA), the APHP-SU R3S (Restoration, Resuscitation, Rehabilitation, Sleep) Department, and the Radboud-UMC University Hospital in the Netherlands.
Other methods and devices developed at the NeuPEL laboratory
MyoTone Test - Assessment of myotonia using force relaxation curve modelling
The MyoTone Test was developed in 2008 in order to assess, in less than 10 minutes, the reaction time, the maximum voluntary grip force and myotonia after a submaximal contraction at 70 % of maximum voluntary force. Myotonia is estimated using modelling with a supervised software application.
Gait analysis using accelerometry
Gait analysis using accelerometry is a promising tool in the study of the clinical parameters of gait. Indeed, this technique is able to provide useful information regarding gait characteristics (quantitative parameters) in healthy subjects or subjects with neuromuscular disease. The recordings are performed with a 3-axis accelerometer, which records all centre-of-gravity motion while walking. The analyses are performed using a software application developed in the laboratory that calculates the spatiotemporal gait parameters based on the accelerometer signals.
Grip-Ball - Connected ball to measure and rehabilitate grip strength (patented)
The Grip-Ball is a tool to assess and rehabilitate palm grip strength that can be used by the subjects themselves with a simple compression movement of the ball in their hand. It consists of a flexible plastic ball that is inflatable and airtight, into which pressure and temperature sensors are integrated, with digital functionality (Bluetooth). The results can be recorded and displayed on the local terminal, then later transmitted to a server. The Grip-Ball was developed in partnership with the Troyes Technology University.
ELAPS - System to measure high spatial resolution EMG (patented)
High spatial resolution EMG, based on the use of a multi-electrode system, combined with a spatial filtering procedure, currently offers the best compromise between the selectivity of the needle and the representativeness of the conventional surface electrode. The system makes it possible to detect the activity of a single motor unit (MU) non-invasively, and to determine the conduction velocity of these motor units. The distribution of conduction velocity is indirectly linked to the size of the muscle fibres. Furthermore, high spatial resolution EMG detects changes in MU electrical activity, typical of certain neuromuscular and neurological diseases.
Assessment of assistance devices – AssistMyo
 We are evaluating the short-term and long-term safety, feasibility and efficacy of innovative augmentation/compensation devices in patients with neuromuscular disease.
We are evaluating the short-term and long-term safety, feasibility and efficacy of innovative augmentation/compensation devices in patients with neuromuscular disease.
Motorised assistance devices, collectively referred to as “exoskeletons”, have a significant potential to compensate for muscle weakness, thus improving patient mobility and independence, outside of the context of rehabilitation.
Exercise and physical activity in the neuromuscular diseases – ExMyo
 We are studying the effects of exercise-based interventions on neuromuscular function. We are also studying the impact of neuromuscular disorders on daily physical activity, depending on the disease and clinical variables. We are using objective physical activity quantification methods, such as accelerometry, combined with innovative approaches involving signal processing. We are also conducting prospective studies to assess the effects of an increase in daily physical activity and/or structured exercise programmes (strength and/or endurance) in patients with neuromuscular diseases.
We are studying the effects of exercise-based interventions on neuromuscular function. We are also studying the impact of neuromuscular disorders on daily physical activity, depending on the disease and clinical variables. We are using objective physical activity quantification methods, such as accelerometry, combined with innovative approaches involving signal processing. We are also conducting prospective studies to assess the effects of an increase in daily physical activity and/or structured exercise programmes (strength and/or endurance) in patients with neuromuscular diseases.
The current work is being conducted in collaboration with the Department of Neuromyology and the APHP-SU Internal Medicine Department. Members of the laboratory are also significantly involved in developing the guidelines regarding physical activity and exercise in neuromuscular diseases (MEDICOSPORT Health).
Clinical diagnosis activities
Grip Test – Non-ischaemic forearm exercise test to detect patients presenting intolerance to stress
The Grip Test is a test used to help diagnose metabolic muscular diseases. It consists of assessing maximum grip strength and making the patient perform a standardised muscle task. Regular blood draws are used to analyse metabolite levels (including lactate and ammonia) in the blood, which change as a result of the exertion undertaken. The analysis of changes in blood levels helps to guide the diagnosis.
The Grip Test was invented in 1998 and established in 2000 in the Department of Neuromyology to replace the so-called tourniquet test, which can be painful or even dangerous. Since that time, almost 2000 patients have benefited from this test in guiding their diagnosis. Several diagnostic algorithms have been proposed, to detect glycogen storage diseases and pseudometabolic dystrophies. Thus, the Grip Test can detect glycogen storage disease 5 (GSD5, McArdle disease) with a sensitivity of 100 % and a specificity of 99.7 % (Hogrel et al, 2015). Hyperammonaemia is a strong criterion in favour of the diagnosis of glycogen storage disease (Hogrel et al, 2017), since, besides GSD5, it can suggest a diagnosis of other glycogen storage diseases (debranching enzyme deficit (AGL), phosphorylase-b kinase (PHKA1), phosphoglycerate kinase 1 (PGK1), phosphoglucomutase-1 (PGM1)). CPK analysis 24 hours after the test is also informative. A normal Grip Test accompanied by significantly elevated CPK at 24 hours (+300 IU/L) suggests a pseudometabolic dystrophy (mainly LGMD or Becker MD). This work is being conducted in collaboration with the APHP-SU Department of Neuromyology.
Involvement in clinical research
 In close collaboration with the I-Motion Adults Clinical Trials Department, we are involved in a number of clinical trials (natural history studies and therapeutic trials) as sponsor or centre investigator for external sponsors, including start-ups and small and large pharmaceutical companies.
In close collaboration with the I-Motion Adults Clinical Trials Department, we are involved in a number of clinical trials (natural history studies and therapeutic trials) as sponsor or centre investigator for external sponsors, including start-ups and small and large pharmaceutical companies.
Expertise and services
Consultancy activities
The NeuPEL laboratory provides consultancy services to university and industrial clients, including study design and monitoring (assessment methods, outcomes, etc.).
Available equipment and techniques
- Wired (Digitimer) and wireless (Delsys) electromyography
- Motion capture and analysis by inertial units (Opale, APDM)
- Isokinetic dynamometer (Biodex)
- Hand-held and stabilised dynamometry (MicroFet, Lafayette, QMT, devices developed internally)
- Metabolic expenditure – analysis of respiratory gases
- Spirometry and investigation of respiratory muscle strength
- Electric stimulation (Digitimer)
- Magnetic stimulation (Magstim, bobine donut/8, bistim)
- x2 ultrasound (Aixplorer, Supersonic Imagine)
- Assessment of daily physical activity (Geneactiv, Actigraph; AX3, Axivity)
- Force plate (AMTI)
- x3 Rowing Machines
Patents
The innovative approaches developed by the NeuPEL laboratory regularly result in filed patents. To date, a dozen or so innovations have been patented.
Last publications
- Birnbaum S. & Andersen L. Exercise and myasthenia gravis. Int Rev Neurobiol. 2025;183:179-189. doi: 10.1016/bs.irn.2025.04.022. Epub 2025 May 6. PMID: 41101821.
- Missel M, Petersen RH, Secher EL, Medeiros EBZ, Rude K, Højgaard JLS, Viby NE, Birnbaum S, Witting N, Andersen LK. Challenges navigating thymectomy with myasthenia gravis: A qualitative study of patient treatment experiences.Appl Nurs Res. 2025 Jun;83:151961. doi: 10.1016/j.apnr.2025.151961.
- Rauh SS, Baudin PY, Stojkovic T, Birnbaum S, Decostre V, Zanfongnon RL, Fromes Y, Hooijmans MT, Strijkers GJ, Hogrel JY, Olivier S, Marty B, Reyngoudt H. Multi-parametric quantitative MRI of the lower limb muscles in a longitudinal study of limb-girdle muscular dystrophy R9. PLoS One. 2025 Apr 28;20(4):e0321463. doi: 10.1371/journal.pone.0321463.
- Voorn EL, Lucia A, Vissing J; 281st ENMC workshop study group. 281st ENMC international workshop: 2nd ENMC workshop on exercise training in muscle diseases; towards consensus-based recommendations on exercise prescription and outcome measures. Hoofddorp, The Netherlands, 4-6 October 2024. Neuromuscul Disord. 2025 Apr;49:105318. doi: 10.1016/j.nmd.2025.105318.
- Feigean R, Afroun-Roca C, Guerrini C, Souchu J, Fer F, Benveniste O, Bassez G, Hogrel JY, Bachasson D. Key determinants of impaired gait performance in adults with neuromuscular diseases: a multiparametric and multimodal analysis. J Appl Physiol (1985). 2025 Apr 24. doi: 10.1152/japplphysiol.00287.2024. Online ahead of print. PMID: 40272844 Free article.
- El Kaïm A, Fer F, Decostre V, Laforêt P, Hogrel JY. A model to predict the 6-Minute Walk Distance in Pompe disease. J Neuromuscul Dis. 2025 Apr 22:22143602251336661. doi: 10.1177/22143602251336661. Online ahead of print. PMID: 40261290 Free article.
- Naddaf E, Skolka MP, Prokop L, Dimachkie MM, Hogrel JY, Benveniste O, Wang Z, Mandrekar J, West CP, Murad MH. A systematic review and meta-analysis of the response to placebo in clinical trials of inclusion body myositis. Rheumatology (Oxford). 2025 Mar 14:keaf146. doi: 10.1093/rheumatology/keaf146. Online ahead of print. PMID: 40085004
- Chesshyre M, Ridout D, Stimpson G, Ricotti V, De Lucia S, Niks EH, Straub V, Servais L, Hogrel JY, Baranello G, Manzur A; UK NorthStar Clinical Network; Muntoni F; iMDEX Network. Dystrophin isoform deficiency and upper-limb and respiratory function in Duchenne muscular dystrophy. Dev Med Child Neurol. 2025 Mar 14. doi: 10.1111/dmcn.16282. Online ahead of print. PMID: 40084496 Free article.
- Tomkinson GR, Lang JJ, Rubín L, McGrath R, Gower B, Boyle T, Klug MG, Mayhew AJ, Blake HT, Ortega FB, International norms for adult handgrip strength: A systematic review of data on 2.4 million adults aged 20 to 100+ years from 69 countries and regions. J Sport Health Sci. 2024 Dec 6;14:101014. doi: 10.1016/j.jshs.2024.101014. Online ahead of print. PMID: 39647778 Free PMC article. Review.
- Moutachi D, Hyzewicz J, Roy P, Lemaitre M, Bachasson D, Amthor H, Ritvos O, Li Z, Furling D, Agbulut O, Ferry A. Treadmill running and mechanical overloading improved the strength of the plantaris muscle in the dystrophin-desmin double knockout (DKO) mouse. J Physiol. 2024 Aug;602(15):3641-3660. doi: 10.1113/JP286425.
- El Kaim A., & Birnbaum S. Les Jeux Paralympiques. Kinesither Rev. 2024, 24(272), 1-3.
- Birnbaum S & Santmarty P. The muscle: an extraordinary organ. Kinesither Rev. 2024, 24(269):1-3.
- van Kooten HA, Horton MC, Wenninger S, Babačić H, Schoser B, Lefeuvre C, Taouagh N, Laforêt P, Segovia S, Díaz-Manera J, Claeys KG, Mongini T, Musumeci O, Toscano A, Hundsberger T, Brusse E, van Doorn PA, van der Ploeg AT, van der Beek NAME; European Pompe Consortium study group on outcome measures. Improving outcome measures in late onset Pompe disease: Modified Rasch-Built Pompe-Specific Activity scale. Eur J Neurol. 2024 Dec;31(12):e16397. doi: 10.1111/ene.16397. Epub 2024 Aug 28. PMID: 39205420 Free PMC article.
- Decostre V, De Antonio M, Servais L, Hogrel JY. Relationship Between Hand Strength and Function in Duchenne Muscular Dystrophy and Spinal Muscular Atrophy: Implications for Clinical Trials. J Neuromuscul Dis. 2024;11(4):777-790. doi: 10.3233/JND-230182. PMID: 38788084 Free PMC article.
- Reyngoudt H, Baudin PY, Caldas de Almeida Araújo E, Bachasson D, Boisserie JM, Mariampillai K, Annoussamy M, Allenbach Y, Hogrel JY, Carlier PG, Marty B, Benveniste O. Effect of sirolimus on muscle in inclusion body myositis observed with magnetic resonance imaging and spectroscopy. J Cachexia Sarcopenia Muscle. 2024 Jun;15(3):1108-1120. doi: 10.1002/jcsm.13451. Epub 2024 Apr 13. PMID: 38613252 Free PMC article. Clinical Trial.
- Traoré M, Noviello C, Vergnol A, Gentil C, Halliez M, Saillard L, Gelin M, Forand A, Lemaitre M, Guesmia Z, Cadot B, Caldas de Almeida Araujo E, Marty B, Mougenot N, Messéant J, Strochlic L, Sadoine J, Slimani L, Jolly A, De la Grange P, Hogrel JY, Pietri-Rouxel F, Falcone S. GDF5 as a rejuvenating treatment for age-related neuromuscular failure. Brain. 2024 Nov 4;147(11):3834-3848. doi: 10.1093/brain/awae107. PMID: 38584513
- Woodcock IR, Tachas G, Desem N, Houweling PJ, Kean M, Emmanuel J, Kennedy R, Carroll K, de Valle K, Adams J, Lamandé SR, Coles C, Tiong C, Burton M, Villano D, Button P, Hogrel JY, Catling-Seyffer S, Ryan MM, Delatycki MB, Yiu EM. A phase 2 open-label study of the safety and efficacy of weekly dosing of ATL1102 in patients with non-ambulatory Duchenne muscular dystrophy and pharmacology in mdx mice. PLoS One. 2024 Jan 25;19(1):e0294847. doi: 10.1371/journal.pone.0294847. eCollection 2024. PMID: 38271438 Free PMC article. Clinical Trial.
- Milev E, Selby V, Wolfe A, Rohwer A, Tillmann R, Ramsey D, Iodice M, Hogrel JY, Baranello G, Scoto M, Muntoni F. Assessment of the upper limb function, strength, and mobility in treatment-naive children with spinal muscular atrophy Types 2 and 3. Muscle Nerve. 2024 Mar;69(3):340-348. doi: 10.1002/mus.28041. Epub 2024 Jan 18. PMID: 38238963

